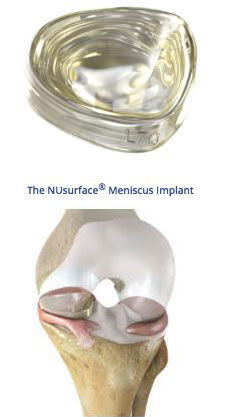
 Patients who have had meniscus knee surgery at least six (6) months prior and are still in pain, may also have hope. Clinical trials are currently being conducted on post surgery patients, from 30 to 75 years old, who are still experiencing knee pain from meniscus deficiency. Previously, braces and meniscal transplants were the only options, other than experimental unconventional therapies. Sites in Boston, Albany, Rochester, New York, Richmond, Memphis, Columbus, Indianapolis, and Chapel Hill are welcoming patients. Those patients receive either traditional post surgical support or a NuSurface meniscus implant. Two hundred and thirty (230) patients will participate. One hundred and forty (140) are already enrolled. Although the NuSurface meniscus implant is an investigational device, it’s manufacturers have been granted an exemption approved by the FDA for the purpose of these trials.
Patients who have had meniscus knee surgery at least six (6) months prior and are still in pain, may also have hope. Clinical trials are currently being conducted on post surgery patients, from 30 to 75 years old, who are still experiencing knee pain from meniscus deficiency. Previously, braces and meniscal transplants were the only options, other than experimental unconventional therapies. Sites in Boston, Albany, Rochester, New York, Richmond, Memphis, Columbus, Indianapolis, and Chapel Hill are welcoming patients. Those patients receive either traditional post surgical support or a NuSurface meniscus implant. Two hundred and thirty (230) patients will participate. One hundred and forty (140) are already enrolled. Although the NuSurface meniscus implant is an investigational device, it’s manufacturers have been granted an exemption approved by the FDA for the purpose of these trials.
Active Implants is in Memphis, Tenn, today. All of the East Coast sites where the trials are being conducted are headed by SportsMed docs. A few surgeons involved, include: Jack Farr in Indianapolis; Brian McKean in Boston; Elliot Hershman in New York; and Chris Kaeding in Columbus at Ohio State. A.I. is not opening more sites and they are at the tail end of patient recruitment.
Stay tuned.
Source:- https://activeimplants.com/products/nusurface-meniscus-implant/















